July 3, 2015
Looking Inside the Biotech Black Box (Part 12)
Some pharmaceutical industry history (1)
The success rate for new drug development is low – about 10% of drugs which enter the clinic are approved. However, regulatory approval does not guarantee market success and only a few novel drug development companies have made, or are likely to make, the transition to top-tier, profitable and sustainable entities which market their own drugs in competition with pharma companies. There are some exceptions such as Amgen, Biogen, Gilead, Regeneron and Celgene. Most companies choose either to license their products to larger pharma companies, generally during the clinical development program, or be acquired. Since the larger pharma companies play such an important role in the fate of the smaller companies, it is important to understand both the history and current status of the pharmaceutical industry.
The modern pharmaceutical industry started in the 1800s as the patent medicine industry, which sold a lot of herb (including cocaine) and alcohol combinations. In the late 1800s and early 1900s, the chemical industry entered the sector, manufacturing natural products such as vitamins and acetylsalicylic acid, for which the original brand product was Aspirin. Various biological products were added through the 1950s, including penicillins, insulin, early vaccines and human blood fractions. The first products based on scientific drug design emerged in the late 1950s and early 1960s including Valium (diazepam), Haldol (haloperidol) and Inderal (propranolol). The birth defects caused by thalidomide in the late 1950s triggered major changes to drug regulation, especially safety testing prior to regulatory approval.
The blockbuster drug strategy emerged in the early 1980s, with the first drug to reach $1 billion in annual sales being Tagamet (cimetidine) for the treatment of excess gastric acid (ulcers). It was developed by Smith, Kline & French (SK&F) and launched in the U.K. in 1976 and in the U.S. in 1979.
 The blockbuster drug strategy emerged in the 1980s and started to show its impact on the share prices of most major drug companies between 1985 and 1990 (easily seen from long term share price charts on a few major drug companies). Before the blockbuster era, the product portfolio of a major drug company would include a small number of unique drug products, many branded generic products (U.S. Hatch-Waxman legislation on bioequivalent generics was only approved in 1984), OTC products and perhaps some vaccine, animal health or diagnostic products. Since the primary focus of the C-level team and board of any public company is the share price and dividend, drug companies began to focus on developing products which could become blockbusters and selling or shutting down product lines with the lowest growth potential.
The blockbuster drug strategy emerged in the 1980s and started to show its impact on the share prices of most major drug companies between 1985 and 1990 (easily seen from long term share price charts on a few major drug companies). Before the blockbuster era, the product portfolio of a major drug company would include a small number of unique drug products, many branded generic products (U.S. Hatch-Waxman legislation on bioequivalent generics was only approved in 1984), OTC products and perhaps some vaccine, animal health or diagnostic products. Since the primary focus of the C-level team and board of any public company is the share price and dividend, drug companies began to focus on developing products which could become blockbusters and selling or shutting down product lines with the lowest growth potential.
Many important new drug products were developed after the adoption of the blockbuster focus. Internal R&D budgets escalated, with the establishment of research facilities which were essentially internal universities. The prices of the new drugs also escalated – as a new stock analyst in 1992, I was very surprised to see the new cancer drug Taxol with a $10,000 annual price tag. Despite the massive increases in R&D budgets, there was no increase in the success rate for clinical development of new drugs.
How can a company replace a drug with $3 billion in sales when patent-based market exclusivity expires and ten generic products are launched the next day at prices which are less than 10% of the brand price? Before we look at the many strategic options for dealing with such losses, we will take a look at patents and market exclusivity in the next blog.
Note: as a follow-up to our previous blogs on cost-effectiveness, Dr. Bach at the Memorial Sloan Kettering Cancer Center has created DrugAbacus, which takes 54 new drugs for cancer and lets you compare the price the company charges to a value-based price (http://www.drugabacus.org/).
[The author and his immediate family members may have long or short positions in the shares of some companies mentioned in or assessed during the preparation of this blog. Past share price performance may not be an indicator of future share price performance. This blog does not consider the investment objectives, financial situation or particular needs of any particular person. Investors should obtain professional advice based on their own individual circumstances before making an investment decision.]
As with all our posts, please see our full legal disclaimer.
___________________________________________________________________
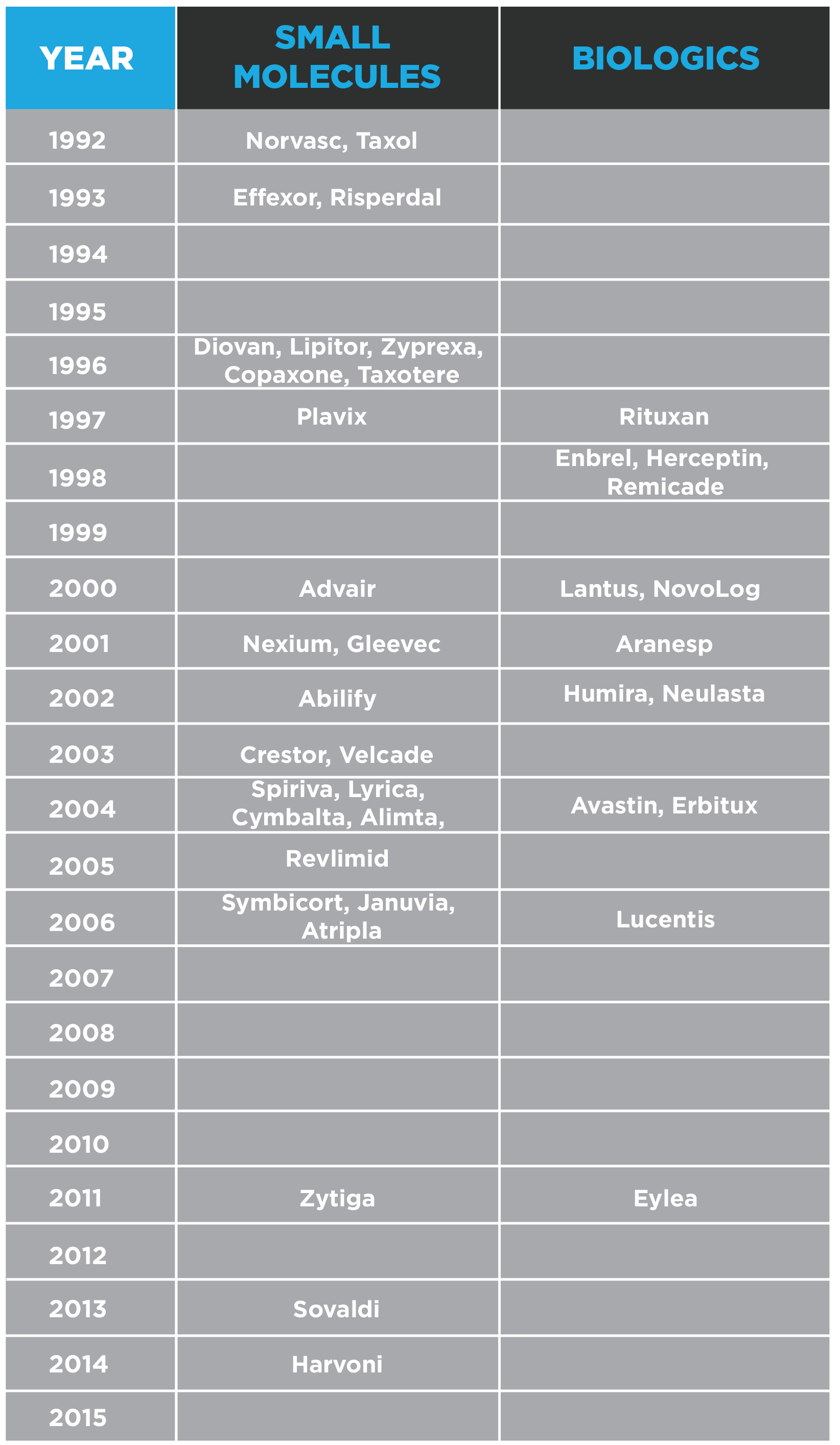
The U.S. approval dates of some of the recent and current blockbusters are shown below. A list of annual New Molecular Entity (NME) and Biologic FDA approvals since 1999 can be found here.